Kidney Dysfunction After Vascularized Composite Allotransplantation.
Authors
Krezdorn N, Tasigiorgos S, Wo L, Lopdrup R, Turk M, Kiwanuka H, Ahmed S, Petruzzo P, Bueno E, Pomahac B, Riella LV.
Abstract
BACKGROUND:Kidney dysfunction is a major complication after nonrenal solid organ transplants. Transplantation of vascularized composite allografts (VCA) has yielded successful midterm outcomes despite high rates of acute rejection and greater requirements of immunosuppression. Whether this translates in higher risks of kidney complications is unknown.METHODS:Ninety-nine recipients of facial or extremity transplants from the Brigham and Women’s Hospital (BWH) and the International Registry on Hand and Composite Tissue Transplantation (IR) were reviewed. We assessed immunosuppression, markers of renal function over time, as well as pretransplant and posttransplant renal risk factors.
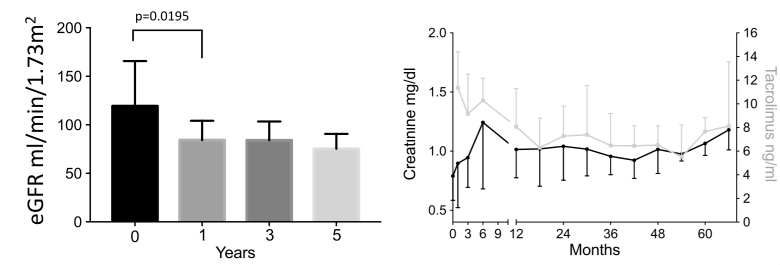
RESULTS:Data were obtained from 10 patients from BWH (age at transplant, 42.5 ± 13.8 years) and 89 patients (37.8 ± 11.5 years) from IR. A significant rise in creatinine levels (BWH, P = 0.0195; IR, P < 0.0001) and drop in estimated glomerular filtration rate (GFR) within the first year posttransplant was observed. The BWH and IR patients lost a mean of 22 mL/min GFR and 60 mL/min estimated GFR in the first year, respectively. This decrease occurred mostly in the first 6 months posttransplant (BWH). Pretransplant creatinine levels were not restored in either cohort. A mixed linear model identified multiple variables correlating with renal dysfunction, particularly tacrolimus trough levels.
CONCLUSIONS:Kidney dysfunction represents a major complication posttransplantation in VCA recipients early on. Strategies to mitigate this complication, such as reducing calcineurin inhibitor trough levels or using alternative immunosuppressive agents, may improve long-term patient outcomes. Standardizing laboratory and data collection of kidney parameters and risk factors in VCA patients will be critical for better understanding of this complication.
Transplant Direct. 2018 Jun 1;4(7):e362.
Figure: Renal function as measured in creatinine levels (mg/dL) and MDRD eGFRs (mL/min per 1.73 m2) over time in the cohort of the BWH