The Notch signaling pathway is known to play a pivotal role in T- and B-cell development and fate, presenting it as an attractive therapeutic target in alloimmunity. This review provides an overview of the mechanisms of Notch signaling, focusing on new insights into its diverse functions in T-cell activation, differentiation and memory subset formation, and the consequences thereof in transplantation.
RECENT FINDINGS:
Recent evidence has shown that while not critical for early antigen-specific CD4 T-cell activation, Notch signaling regulates the survival of memory CD4 T cells via control of glycolytic metabolism; in contrast, Notch signaling is critical for the generation of short-lived CD8 effector T cells, but not memory CD8 cells. Transient, selective inhibition of various Notch receptors and ligands in models of solid organ transplantation has been shown to successfully modulate the alloimmune response, affecting the balance between effector and regulatory cells, with particular influence on the natural regulatory T-cell population.
SUMMARY:
These studies reveal diverse roles for individual Notch receptors and ligands in peripheral immunity and indicate that selective targeting of the Notch pathway is a promising, novel approach for immune modulation in transplantation; the advent of therapeutic human antibodies to neutralize both the Notch ligands and the individual Notch receptors suggests that this approach could be efficiently developed.
Link to video summarizing this review paper: https://vimeo.com/142528215
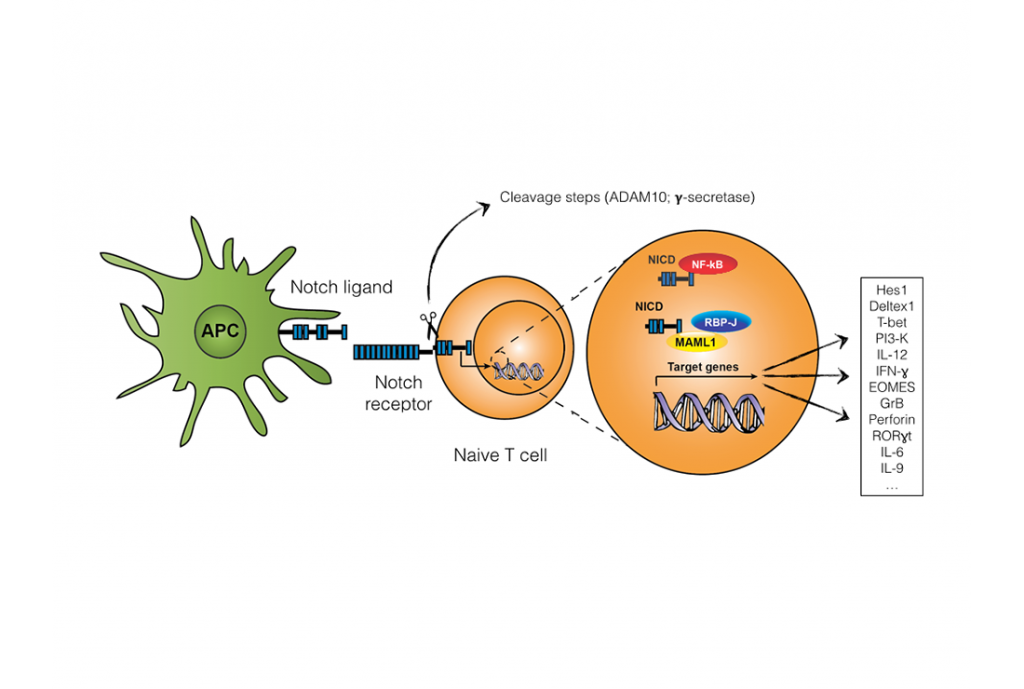
Notch signaling pathway. Notch receptors reside on the cell surface as noncovalently linked heterodimers that are comprised of the extracellular and transmembrane (intracellular) Notch polypeptides. The interaction with Notch ligand induces proteolytic cleavage of the Notch receptor by sequential cleavages mediated by a disintegrin and metalloproteinase and subsequently by gamma-secretase complex. This results in the release of the Notch intracellular domain from the plasma membrane and its translocation to the nucleus, where it binds to recombination-signal-binding protein for immunoglobulin-kJ region transcription factors and coactivators such as Mastermind-like 1 to activate the expression of target genes (limited list of genes depicted on figure). Notch intracellular domain can also exert its function independent of recombination-signal-binding protein for immunoglobulin-kJ through binding to NF-kB (noncanonical pathway).